The realm of single-atom catalysis is rapidly evolving, demonstrating extraordinary potential for enhancing catalytic processes. A recent pioneering study led by Professor Yan Wensheng from the University of Science and Technology of China (USTC) has shed light on a complex phenomenon known as the “volcano-type” relationship. This relationship intricately links metal loading to the activity of the acidic oxygen evolution reaction (OER). By advancing our comprehension of how catalysts behave at the atomic level, this research offers promising pathways for future innovations in energy applications.
The Challenge of Metal Loading
One of the most significant hurdles in single-atom catalysis lies in the fabrication of metal catalysts that can achieve substantial metal loading while maintaining atomic-level dispersion. This is critical, as the efficiency of many catalytic processes hinges on the active site density. The findings presented by Prof. Yan’s team highlight the importance of understanding the balance between metal loading and catalytic performance. By employing a straightforward P-anchoring strategy, they successfully synthesized a series of iridium (Ir) single-atom catalysts with metal loadings between 5% and 21wt%. Interestingly, this innovative approach not only facilitated the maintenance of atomic dispersion but also unveiled critical insights into the inherent dynamics of metal interactions at elevated loadings.
Insights from Advanced Spectroscopy Techniques
Utilizing advanced techniques such as synchrotron radiation X-ray absorption spectroscopy (XAS) and X-ray photoelectron spectroscopy (XPS), the research team elucidated the role of the Ir-P coordination structure. This stable coordination is key to preventing the aggregation of Ir atoms at high loadings, effectively enhancing their interaction efficiencies. The brilliance of this finding lies in its dual nature—the coordination structure not only stabilizes the metal but also significantly impacts the overall catalytic behavior.
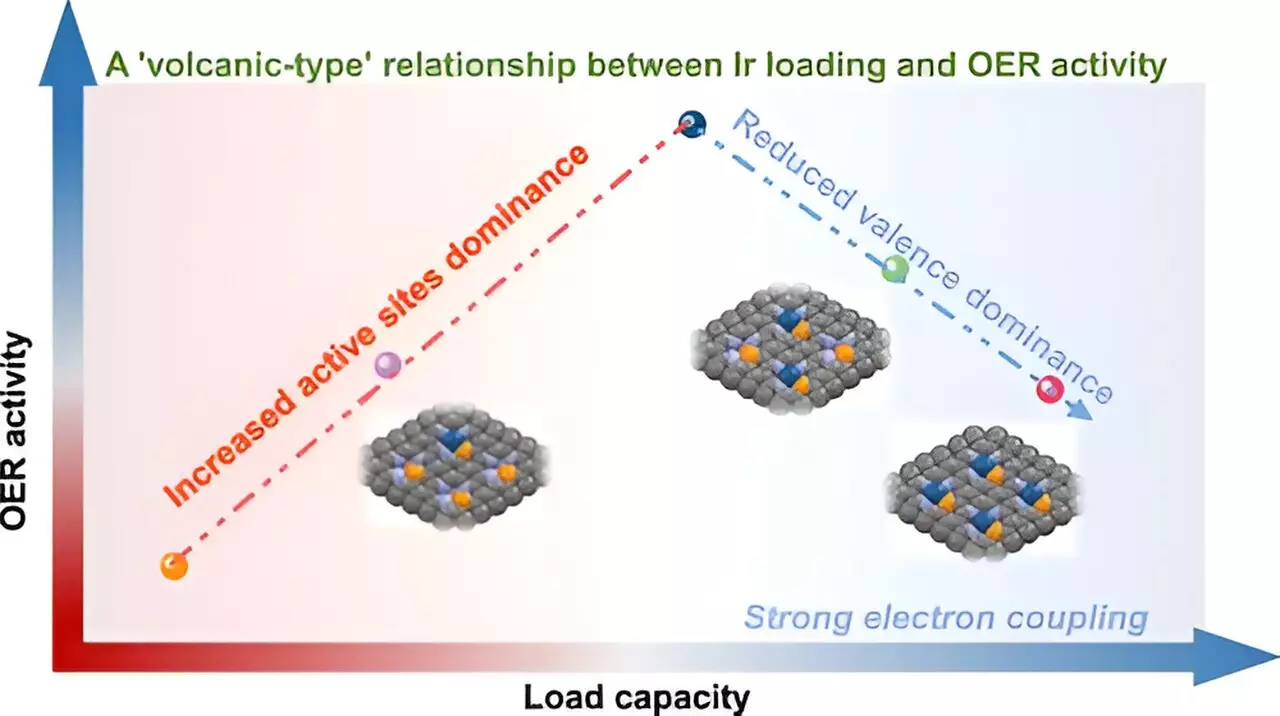
The “Volcano-Type” Phenomenon
The core revelation of this study hinges on the nuanced understanding of the volcano-type relationship. Contrary to traditional assumptions of a linear correlation between metal loading and catalytic activity, the findings indicate a more complex interplay. As Ir loading increases, an initial boost in catalytic activity occurs due to the increase in available active sites. However, surpassing a threshold leads to an intensified interaction between adjacent Ir atoms, negatively impacting their valence state and overall OER activity. This non-linear relationship serves as a critical reminder that in catalysis, more is not always better.
Implications for Future Research and Development
The present study provides a valuable theoretical foundation for the optimization of single-atom catalysts in a variety of applications, particularly in energy conversion and storage technologies. By intricately unraveling the molecular dynamics at play, this research sets the stage for developing more efficient, economically viable catalysts that can meet the pressing demands of energy sustainability. The innovative approaches discussed remind us of the endless possibilities that lie ahead in the pursuit of more effective catalytic systems. As we progress, it’s crucial to continue exploring the delicate dance between atomic structure and catalytic performance, ensuring that our strategies are finely tuned to harness the full potential of these groundbreaking materials.