The escalating crisis of plastic pollution has reached alarming levels, with estimates suggesting that around 8.3 billion metric tons of plastics have been produced worldwide since the mid-20th century. This staggering figure coincides with a disheartening reality: approximately 6.7 billion metric tons of these plastics remain in landfills or have been incinerated, creating a severe environmental burden. Only a meager fraction—600 million metric tons—has been recycled successfully, prompting an urgent call for innovative methods that can effectively address the recycling of unsaturated polymers like rubber and plastics, which dominate our waste streams.
Pioneering Research in Polymer Science
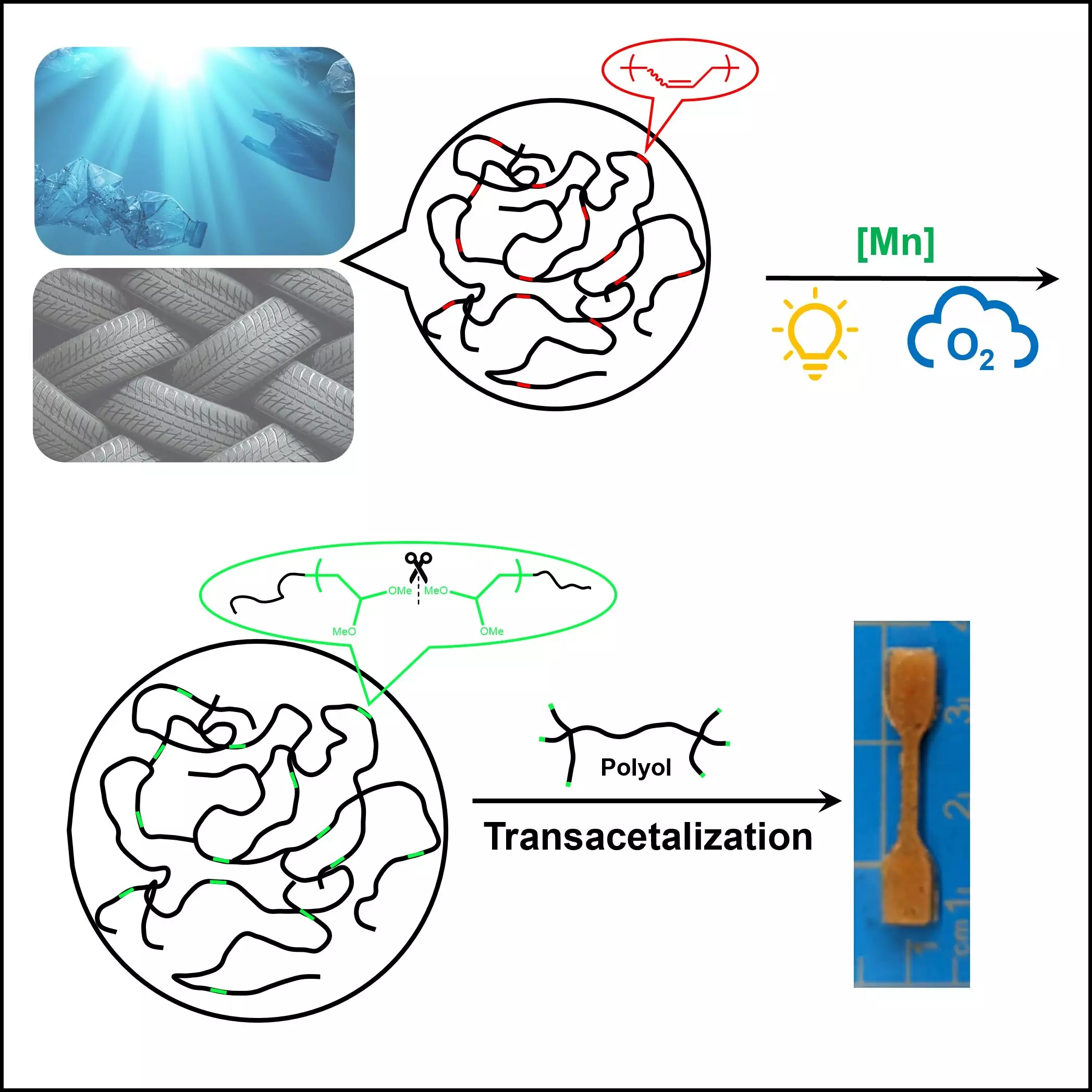
Recent advancements led by Dr. Junpeng Wang and a dedicated team from The University of Akron herald a breakthrough in this field. Their study, featured in Cell Reports Physical Science, reveals a novel approach that leverages the power of oxygen and light to decompose unsaturated polymers. For decades, the inherent stability and durability of commercial polymers, particularly polyolefins, have hindered recycling efforts due to their robust hydrocarbon structures. This innovative research proposes a dynamic technique to introduce unsaturation in these polymers, thereby enhancing their reactivity and creating a pathway for more efficient recycling.
Challenges with Traditional Methods
Traditional recycling techniques often involve energy-intensive processes such as ozonolysis and permanganate oxidation. While these methods may yield results, they are fraught with challenges, including the generation of toxic by-products and a dependence on high-energy conditions. These factors complicate scalability and hinder the feasibility of widespread application. The environmental ramifications of these conventional methods only serve to reinforce the need for greener alternatives in polymer recycling.
A Breakthrough Methodology
What sets the research by Wang and his team apart is the development of a controlled, efficient method that utilizes a light-activated catalyst to break down polymers at room temperature. This significant reduction in energy requirements not only aligns with sustainability goals but also showcases the potential for adopting this technology in a scalable manner. The use of O2 as an oxidizing agent, from both an environmental perspective and its abundance, presents a game-changing opportunity in the polymer recycling landscape.
Implications for the Future of Recycling
This groundbreaking work represents more than just an academic achievement; it presents a transformative approach to an industry burdened by inefficiency and pollution. By enhancing our grasp of polymer degradation, Wang and his collaborators are laying the groundwork for a sustainable future where unsaturated polymers can be effectively recycled, thereby contributing to a circular economy. This research conveys a powerful message: with innovation and scientific inquiry, we can collaboratively tackle one of the pressing environmental issues of our time.