In the ever-evolving world of organic chemistry, the search for more efficient and selective methodologies is relentless. A significant leap in this direction has been achieved by Professor Max Martin Hansmann and his research team, who have ingeniously developed a new reagent capable of adding carbon atoms to various molecular structures selectively and efficiently. This breakthrough, substantiated by a robust study published in *Science*, is poised to reshape the fundamental landscape of organic synthesis, particularly in the realm of drug discovery and complex molecule construction.
The critical essence of this advancement lies not only in its synthetic utility but also in its conceptual framework. For years, chemists have grappled with the dual challenges of achieving selectivity and efficiency in carbon transfer reactions—a feat that remains daunting but essential for developing complex pharmaceuticals. The innovative work by Hansmann’s team not only targets these challenges head-on but also offers a pathway toward synthesizing sophisticated molecular constructs with fewer synthetic steps, thereby reducing time and resource expenditure.
Unveiling the Carbon Source: A Novel Reagent
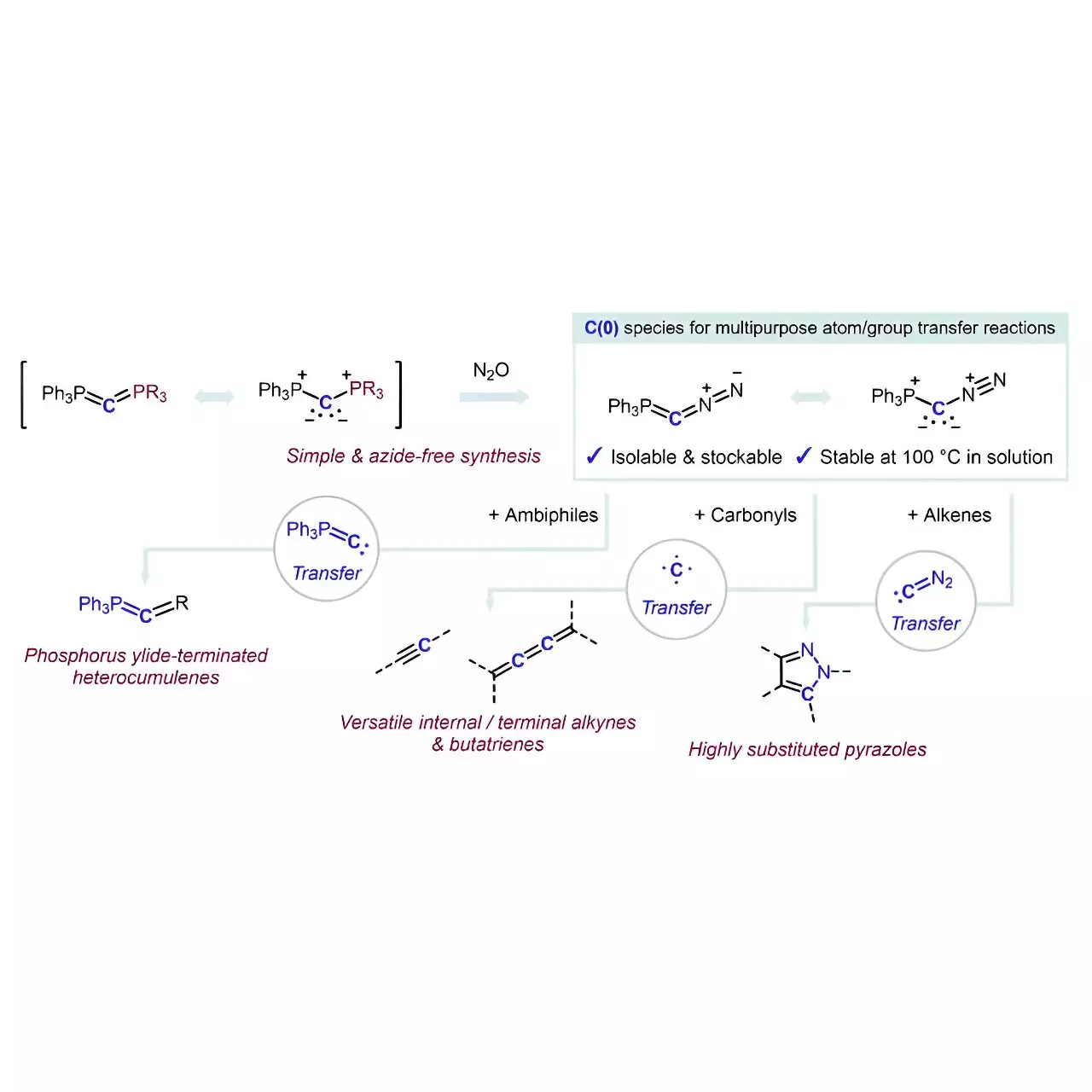
At the heart of this remarkable advancement is the development of a reagent that exemplifies both a carbon atom source and a multipurpose transfer reagent. This dual capability positions the reagent as an elegant solution to a long-standing issue in organic chemistry. The new approach involves a stable carbon species known as carbones, which have scarcely been leveraged in previous research due to inherent complexities and challenges associated with their use.
Professor Hansmann and his team succeeded in stabilizing a carbon atom through strategic coordination with two neutral, electron-donating groups. The meticulous design and synthesis of the crystalline reagent, Ph3P=C=N2, not only streamline the carbon atom’s accessibility but also eliminate reliance on azides—compounds associated with significant safety risks. By employing a straightforward PPh3/N2 exchange reaction with carbodiphosphorane and nitrous oxide, the team developed a reagent that is both elegant and fundamentally simpler.
Applications and Implications
The implications of this work are far-reaching, opening doors to unforeseen applications in organic synthesis and pharmaceutical development. Hansmann expresses optimism that the newly synthesized reagent will catalyze innovative methodologies in carbon atom transfer—potentially redefining how researchers approach the construction of higher cumulenes and facilitating late-stage functionalization of complex molecular architectures.
For instance, their findings illustrate that the reagent facilitates specific carbon atom transfers leading to the formation of various alkynes and butatrienes from carbonyl compounds. The versatility offered by this reagent is crucial, as it promises greater accessibility to otherwise intricate molecular pathways that were once considered overly complex or inefficient to pursue.
Furthermore, the practical applications extend to the generation of phosphorus ylide-terminated heterocumulenes and multi-substituted pyrazoles. The ability to accomplish these transformations without additional additives enhances the reagent’s practicality and enhances the efficiency of the overall synthetic process.
A Bright Future for Organic Chemists
The potential this reagent holds is a breath of fresh air in the field of organic chemistry, filled with hope for chemists striving for more efficient paths to complex molecules. As the research progresses, it is likely that the carbon transfer reagent will become a staple tool in the chemist’s toolkit, facilitating collaborations and innovations across pharmaceutical chemistry, materials science, and beyond.
Research teams worldwide will undoubtedly monitor Hansmann’s work closely, as the strategies exhibited here could lay the groundwork for future advancements. The scientific community stands at the precipice of a new frontier, thanks to the visionary endeavors of Professor Hansmann and his dedicated team. With further exploration and understanding of this reagent’s reactivity, we can anticipate a flourishing era of synthetic chemistry that leverages precision and innovation to unlock complex pharmaceuticals in ways we have only begun to imagine.