Nitrate contamination in water sources poses a significant challenge to both environmental integrity and public health. Although nitrates are naturally occurring in soil and water due to agricultural runoff and fertilizer use, their excessive levels can lead to severe health issues, including methemoglobinemia, or “blue baby syndrome,” which affects infants’ ability to transport oxygen. Additionally, high nitrate levels can harm aquatic ecosystems, resulting in algae blooms that deplete oxygen levels in water bodies. Traditional methods of remediation often fall short, leading to a pressing need for innovative solutions.
A Breakthrough in Purification Technology
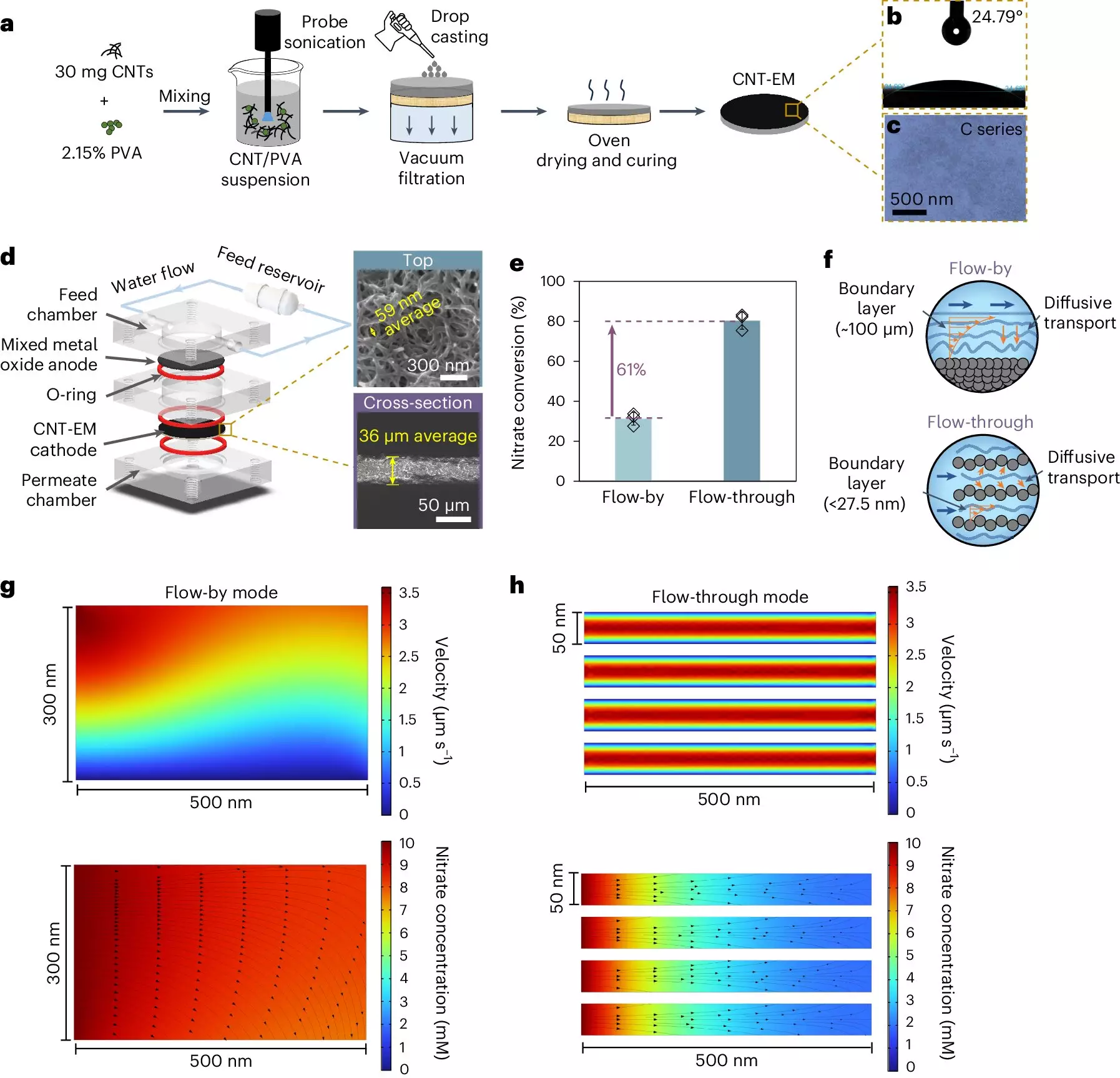
In light of these pressing concerns, researchers at Yale University have introduced a groundbreaking method for efficiently removing nitrates from drinking water. Spearheaded by Lea Winter, an assistant professor in chemical and environmental engineering, the new approach utilizes electrified membranes crafted from carbon nanotubes. This innovative technology not only simplifies the nitrate removal process but also mitigates the limitations seen in conventional methods.
As Winter highlights, existing techniques generally rely on either separating the nitrates from water or degrading them. The separation process often results in concentrated waste that risks re-entering the ecosystem, which can be counterproductive. Hence, a more viable strategy is the destruction of nitrates themselves. Traditional biological denitrification methods used in wastewater treatment plants are slow, sensitive to environmental changes, and require microbial intervention, which can be temperamental.
The Electrocatalytic Advantage
A significant advancement in Winter’s methodology is the use of electrocatalytic processes. Unlike the slower biological processes, electrocatalysis allows for efficient nitrate destruction, providing more control over the purification process. However, conventional electrochemical systems face challenges related to their design. For instance, the two-dimensional flat plate electrodes commonly used hinder the flow of nitrates toward the reaction sites, resulting in slow reaction kinetics.
Winter’s lab addresses this limitation by employing electrified membranes with exceedingly small pore sizes—about 50 nanometers compared to the standard 100 micrometers seen in classic systems. This innovative design creates a more conducive environment for nitrate transport, significantly enhancing the efficiency of nitrate removal. By bypassing the thick boundary layer that often hampers fluid dynamics in traditional setups, Winter’s team enables rapid nitrate interaction with the electrode surface.
The Non-Metal Catalyst Innovation
Another key innovation with the electrified membranes is the elimination of metal catalysts, which are typically used in electrochemical processes to facilitate nitrate conversion. Instead, Winter’s membranes fully leverage carbon nanotubes as catalysts, achieving nitrate removal rates comparable to those seen with metals but in a fraction of the time. Remarkably, what used to take hours to accomplish in conventional systems can now be achieved in as little as 15 seconds.
This dramatic reduction in processing time is revolutionary not just for the technology itself, but also for the broader implications it holds for water treatment facilities, public health, and environmental protection. The speed of reaction means that facilities can manage large volumes of contaminated water far more efficiently, improving their operational capacity and responsiveness to nitrate pollution.
Real-World Applications and Future Prospects
To validate the practical applicability of this novel technology, Winter’s research team conducted experiments on water samples from Lake Wintergreen, a location near the Yale campus. By introducing controlled nitrate concentrations to these samples, the researchers were able to observe the effectiveness of their carbon nanotube membranes in real-world conditions. Their focus on lower concentration levels—a common scenario in environmental contamination—demonstrates the versatility and potential of this method for various water quality situations faced globally.
The implications of this research extend far beyond laboratory settings. As water scarcity and contamination become increasingly critical issues worldwide, the ability to rapidly reduce nitrate concentrations could lead to profound changes in how we manage and treat our water supplies. The efficiencies gained through this technology may ultimately pave the way for new water purification standards, leading to healthier communities and ecosystems.
Lea Winter’s pioneering work with electrified carbon nanotube membranes is a significant step forward in combating nitrate contamination. By revolutionizing water purification techniques and aligning effectiveness with environmental responsibility, Winter and her team are setting a new standard for future water treatment technologies. This could represent not just a scientific achievement, but a vital breakthrough in global efforts to ensure clean water access for all.