At the forefront of green chemistry, a groundbreaking development has emerged from the research team at UNIST, under the dedicated leadership of Professor Jaeheung Cho. Their creation of a revolutionary catalyst showcasing enzymatic qualities is set to change the landscape of how we combat harmful hydrocarbon pollution. Unlike traditional methods that often require high energy consumption, this innovative catalyst mimics the catalytic processes of metalloenzymes, naturally occurring entities known for their efficiency in breaking down complex organic molecules.
Mechanistic Insights into Catalytic Efficiency
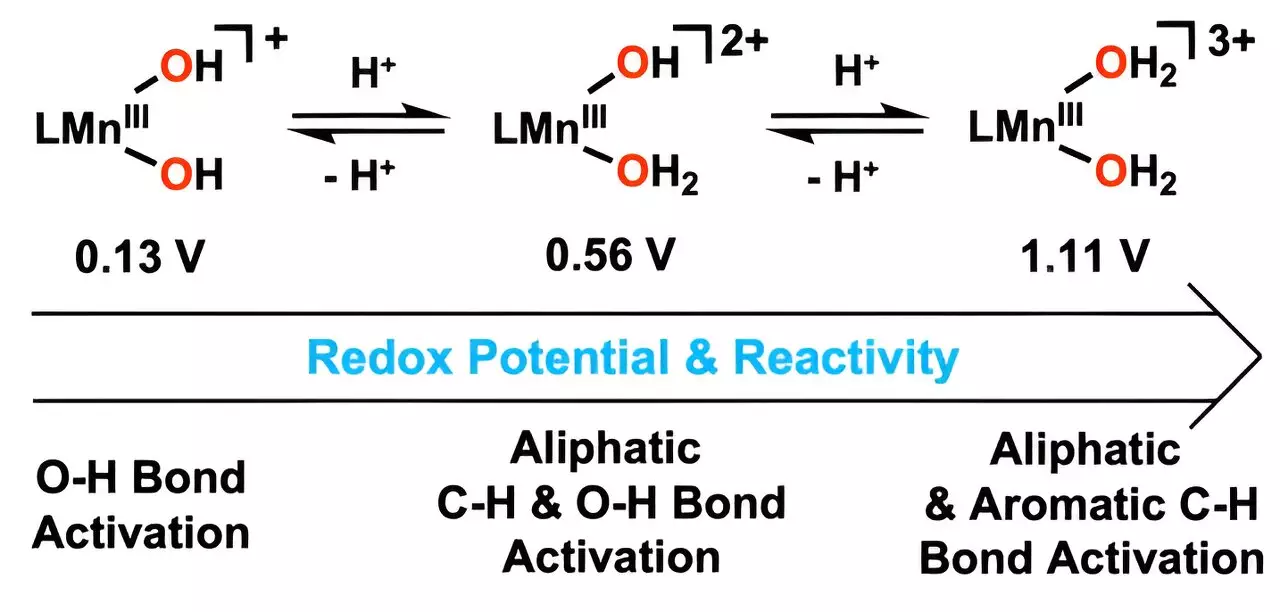
What makes this advancement particularly compelling is the catalyst’s mechanism of action, which involves the incorporation of hydrogen ions into hydroxo ligands. This strategic modification enables the synthesis of metal-bound water molecules, allowing the catalyst to facilitate the oxidation of robust carbon-hydrogen (C–H) bonds at significantly lower temperatures. This is not merely a minor improvement; it signals a fundamental shift in catalysis that could pave the way for more sustainable industrial applications.
Professor Cho points out that the catalyst’s performance has improved dramatically due to these structural adjustments, particularly within the manganese component. The increase in the reduction potential allows for greater activation of oxygen-hydrogen bonds, leading to accelerated reaction rates. This means that chemical processes can be executed more efficiently and with less energy input, a critical aspect in the fight against climate change.
Real-World Implications and Applications
The implications of this research extend far beyond academic significance. The capability of the new catalyst to efficiently oxidize anthracene, a challenging substance known for its strong carbon-hydrogen bonds, showcases its practical application potential. Furthermore, its ability to decompose aromatic hydrocarbons that resist traditional degradation methods compounds its importance. These properties suggest that this catalyst could be instrumental in environments where oil spills and other hydrocarbon pollutants pose serious ecological threats.
This achievement not only highlights the sophisticated interplay between metal chemistry and environmental science but also illustrates the immense potential for such technologies in industrial settings. The emphasis on low-temperature operations suggests that energy costs associated with chemical reactions could be drastically reduced, which is crucial for industries aiming for greener processes. With control over the manganese’s reduction potential, there is an exciting trajectory to develop metal catalysts that hold the promise of revolutionizing pollution control.
Towards a Sustainable Industrial Future
The advancements pioneered by Professor Cho’s team not only open new avenues for research but also inspire hope for the future of sustainable chemistry. By harnessing the power of nature’s own mechanisms, this novel catalyst lays the groundwork for reducing harmful emissions and minimizing our environmental footprint. As industries increasingly seek to adhere to regulatory pressures and societal demands for sustainability, innovations like these offer viable solutions. The emphasis on environmentally friendly, energy-efficient technology is not just a trend but a necessity for the planet’s future. This catalyst epitomizes how scientific ingenuity can provide us with tools to address some of the most pressing challenges of our time.